 
However, the arrangement of the bonds is the reason, which creates a barrier in front of SiF4 for being polar compound. The high polarity between the bonds made by Silicon and Fluorine can be a great reason for considering the compound as a polar compound. That reason is considered as the strong fact for SiF4 to possess zero dipole moment even after having polar bonds. There is a huge reason, which generates complete non-polar nature among Silicon tetrafluoride. Otherwise, the dipole nature of the bonds is very strong. Silicon tetrafuloride has polar bonds between Silicon and fluorine but the tetrahedral shape eliminates the dipole movement of four Si-F sigma bonds. The compound occurs as great example of non-polar compound, which is quite attainable for showing the exceptional compound in the series of the polar compounds. The shape and the bond structure of the compound give reasonable approaches to provide proper consideration about the polarity of SiF4. There is a huge contrary explanation about the fact regarding polar non-polar nature of SiF4, Silicon tetrafluoride. Lewis dot structure of the compound would be relevantly presented in this article as well. This article would represent the chemistry behind considering the polarity of SiF4. It does not store any personal data.“Is SiF4 polar or nonpolar” is the main descriptive topic in this article. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". 
This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Therefore, the Dipole moment of XeF2 is 0 D. The dipole moment of a molecule is the measure of its polarity. SF4 has sp2d2-hybridization and see-saw goemetry.ĭue to the difference between the electronegativity of fluorine and xenon atom, the molecule of XeF2 ensures non zero dipole moment originating in the direction of fluorine. The molecule having permanent dipole moment is. The covalent molecule BeCl2 has permanent dipole moment. Hence, the molecule is polar with non zero dipole moment. Hence, the molecule is a polar molecule and has a dipole moment. Hence, the chlorine-fluorine bond is polar, and has a dipole moment. Because carbon and hydrogen in benzene molecules have distinct electronegativities, the molecule has a symmetrical planar structure. Asymmetric or different electro-negativities characterize molecules with a dipole moment. What is the dipole moment of benzene?īenzene has a dipole moment of zero. Due to asymmetric molecular shape, the distribution of charges across the molecules of SF4 is uneven. 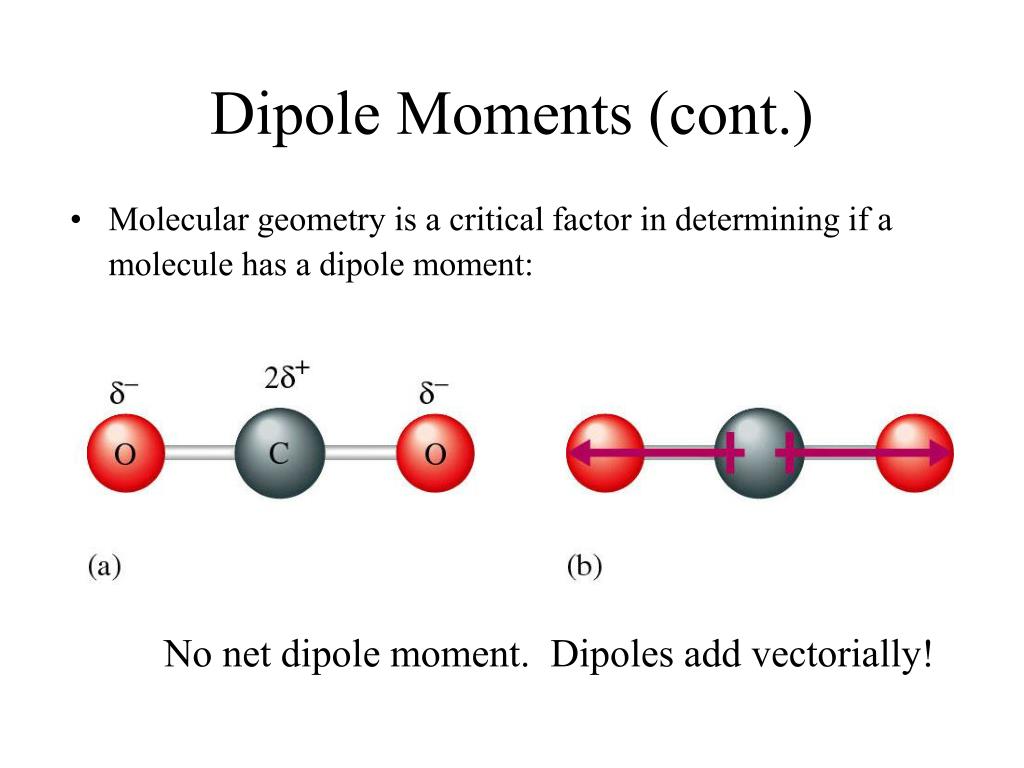
Moreover, Fluorine is more electronegative than Sulfur due to which the overall charge distribution of a molecule is uneven resulting in a polar molecule and give 0.632 D dipole moment. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
